Tadalafil and analogues
Abstract
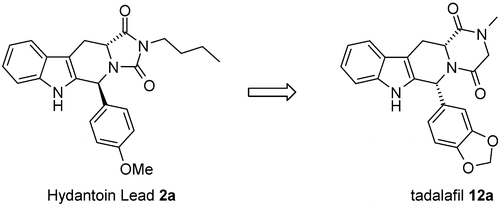
Modification of the hydantoin ring in the previously described lead compound 2a has led to the discovery of compound 12a, tadalafil, a highly potent and highly selective PDE5 inhibitor. The replacement of the hydantoin in compound 2a by a piperazinedione ring led to compound cis-11a which showed similar PDE5 inhibitory potency. Introduction of a 3,4-methylenedioxy substitution on the phenyl ring in position 6 led to a potent PDE5 inhibitor cis-11c with increased cellular potency. Optimization of the chain on the piperazinedione ring led to the identification of the racemic cis-N-methyl derivative 11i. High diastereospecificity for PDE5 inhibition was observed in the piperazinedione series with the cis-(6R,12aR) enantiomer displaying the highest PDE5 inhibitory activity. The piperazinedione 12a, tadalafil (GF196960), has been identified as a highly potent PDE5 inhibitor (IC50 = 5 nM) with high selectivity for PDE5 vs PDE1−4 and PDE6. Compound 12a displays 85-fold greater selectivity vs PDE6 than sildenafil 1. 12a showed profound and long-lasting blood pressure lowering activity (30 mmHg/>7 h) in the spontaneously hypertensive rat model after oral administration (5 mg/kg).